Testing Plant Substances as Potential Medicines
Background: Many plants contain advantageous properties which can be used in the making of medicine. This is evident because plants produce antimicrobial agents which battle the microbes. By isolating the antimicrobial molecule, a potential therapeutic medicine could be created.
Purpose: To create a therapeutic medicine using plant extracts.
Materials List:
-balance, weigh boat, lab scoops -LB broth base
-media bottles, 250mL -sterilizer/autoclave
-water bath, 37°C, shaking -sterile LB agar
-laminar flow hood/disinfectant -plastic safety goggles
-bunsen burner and gas lighter -inoculating loop
-petri dish, 60x15mm, sterile -E. Coli
-plant specimen -mortar and pestle
-pipet, 10mL and pump -plastic funnels, short stem
-filter paper disks, 5mm diameter -beakers, 100mL
-syringe, 10mL and filter 0.2mm -reaction tubes/rack, 1.7mL
-methanol, absolute -pipet, 1mL and pump
-dry heat block -forceps, fine tipped
-ampicillin -glass spreader
-incubator oven, 37°C
Procedure:
1. Prepare a nutrient or LB culture for the E. coli at least 24 hours in advance. Using sterile technique, add a colony of E. coli culture to the broth medium and incubate, shaking at 37 C for 24 hours.
2. Each lab group need 2 petri plates. Draw a "+" on each plate bottom to divide the pate into quadrants (four sections). Label the quadrants No. 1 through 4. Also, label the dish with your initials and the date.
3. Liquefy sterile LB agar in the microwave at 50% power. Using sterile technique, pour approximately 20 mL of sterile, liquid LB agar into each Petri plate. Let the agar solidify for 15 minutes. Let it dry for at least 24 hours.
4. Using a mortar and pestle, grind up 2 g of plant tissue (leaves or bark) with 10 mL of deionized water. Lwt it sit for 3 minutes. Filter and sample through an 11 cm filter paper funnel. Filter sterilize the filtered sample extract using a syringe filter, as demonstrated by the instructor. Collect with 1 mL of extract into a 1.7 mL microtube. Label the sample.
5. Repeat Step 4, but replace the water with methanol as as the extracting solvent. After the methanol extraction, place the 1.7 mL tube with the 1 mL of methanol extract in a 65 C heat block (caps open) for 24 hours or more, if necessary to evaporate the methanol. Reconstitute dry matter in the tube with 1 mL of deionized water.
6. For each of the other samples, repeat steps 4 and 5. Label all samples. There should be six tubes of samples.
7. Using sterile forceps (that have been flamed in alcohol) drop three filter paper disks into each tube of the filtered extract.
8. Prepare negative control disks, three each, of only methanol and only sterile distilled water.
9. Prepare six positive control disks of ampicillin solution.
10. Allow the disks sufficient time to soak up enough extract to be saturated (perhaps overnight).
11. Close the tubes. Store all samples at 4 C until ready to use.
12. Using a sterile pipette transfer 1 mL of the E. coli broth
(made at step 1) to the middle of each Petri dish. Sterilize a spreading loop (using alcohol and a flame) , and evenly spread the bacterial culture around the Petri plate. Quickly cover, and allow the culture to soak into the agar for at least 15 minutes.
13. Using sterile forceps carefully place one disk into the middle of each quadrant, about 2 cm from the outer edge of the Petri dish. Blot any excess liquid before placing the disk on the Petri dish. Keep all the methanol-extracted samples on the same dish and all the water-extracted samples on the same dish.
14. Repeat step 13 twice so that you have three replicates of the methanol extraction and three replicates of the deionized water extractions.
15. Place one of the negative control disks, either sterile distilled water or methanol, in the center of the appropriate plate. Place a positive control disk with ampicillin in another quadrant of each plate.
16. You should end up with six Petri plates, each containing a negative control in the middle, a positive control , and three sample disks. Make sure you have recorded exactly which plant extracts and which solvent went into each quadrant.
17. Make sure the disks are adhering well to the surface of the agar. For incubation, invert the plates and incubate at 37 C for 24 to 48 hours.
18. After incubation, examine the plates with the plant extract disks for zones of inhibition. This is a clear area formed around the disk by the inhibitory action of a substance(s) in the plant material. Photograph or draw the plates, labeling any inhibition of bacterial growth.
19. Create a data table to collect and present data of all the replicates as well as the averages. include descriptions of the bacterial lawn around each disk. Measure and record the diameter and clarity of any cleared areas around the disks. Give quantitative measurements of your observations.
Background: Many plants contain advantageous properties which can be used in the making of medicine. This is evident because plants produce antimicrobial agents which battle the microbes. By isolating the antimicrobial molecule, a potential therapeutic medicine could be created.
Purpose: To create a therapeutic medicine using plant extracts.
Materials List:
-balance, weigh boat, lab scoops -LB broth base
-media bottles, 250mL -sterilizer/autoclave
-water bath, 37°C, shaking -sterile LB agar
-laminar flow hood/disinfectant -plastic safety goggles
-bunsen burner and gas lighter -inoculating loop
-petri dish, 60x15mm, sterile -E. Coli
-plant specimen -mortar and pestle
-pipet, 10mL and pump -plastic funnels, short stem
-filter paper disks, 5mm diameter -beakers, 100mL
-syringe, 10mL and filter 0.2mm -reaction tubes/rack, 1.7mL
-methanol, absolute -pipet, 1mL and pump
-dry heat block -forceps, fine tipped
-ampicillin -glass spreader
-incubator oven, 37°C
Procedure:
1. Prepare a nutrient or LB culture for the E. coli at least 24 hours in advance. Using sterile technique, add a colony of E. coli culture to the broth medium and incubate, shaking at 37 C for 24 hours.
2. Each lab group need 2 petri plates. Draw a "+" on each plate bottom to divide the pate into quadrants (four sections). Label the quadrants No. 1 through 4. Also, label the dish with your initials and the date.
3. Liquefy sterile LB agar in the microwave at 50% power. Using sterile technique, pour approximately 20 mL of sterile, liquid LB agar into each Petri plate. Let the agar solidify for 15 minutes. Let it dry for at least 24 hours.
4. Using a mortar and pestle, grind up 2 g of plant tissue (leaves or bark) with 10 mL of deionized water. Lwt it sit for 3 minutes. Filter and sample through an 11 cm filter paper funnel. Filter sterilize the filtered sample extract using a syringe filter, as demonstrated by the instructor. Collect with 1 mL of extract into a 1.7 mL microtube. Label the sample.
5. Repeat Step 4, but replace the water with methanol as as the extracting solvent. After the methanol extraction, place the 1.7 mL tube with the 1 mL of methanol extract in a 65 C heat block (caps open) for 24 hours or more, if necessary to evaporate the methanol. Reconstitute dry matter in the tube with 1 mL of deionized water.
6. For each of the other samples, repeat steps 4 and 5. Label all samples. There should be six tubes of samples.
7. Using sterile forceps (that have been flamed in alcohol) drop three filter paper disks into each tube of the filtered extract.
8. Prepare negative control disks, three each, of only methanol and only sterile distilled water.
9. Prepare six positive control disks of ampicillin solution.
10. Allow the disks sufficient time to soak up enough extract to be saturated (perhaps overnight).
11. Close the tubes. Store all samples at 4 C until ready to use.
12. Using a sterile pipette transfer 1 mL of the E. coli broth
(made at step 1) to the middle of each Petri dish. Sterilize a spreading loop (using alcohol and a flame) , and evenly spread the bacterial culture around the Petri plate. Quickly cover, and allow the culture to soak into the agar for at least 15 minutes.
13. Using sterile forceps carefully place one disk into the middle of each quadrant, about 2 cm from the outer edge of the Petri dish. Blot any excess liquid before placing the disk on the Petri dish. Keep all the methanol-extracted samples on the same dish and all the water-extracted samples on the same dish.
14. Repeat step 13 twice so that you have three replicates of the methanol extraction and three replicates of the deionized water extractions.
15. Place one of the negative control disks, either sterile distilled water or methanol, in the center of the appropriate plate. Place a positive control disk with ampicillin in another quadrant of each plate.
16. You should end up with six Petri plates, each containing a negative control in the middle, a positive control , and three sample disks. Make sure you have recorded exactly which plant extracts and which solvent went into each quadrant.
17. Make sure the disks are adhering well to the surface of the agar. For incubation, invert the plates and incubate at 37 C for 24 to 48 hours.
18. After incubation, examine the plates with the plant extract disks for zones of inhibition. This is a clear area formed around the disk by the inhibitory action of a substance(s) in the plant material. Photograph or draw the plates, labeling any inhibition of bacterial growth.
19. Create a data table to collect and present data of all the replicates as well as the averages. include descriptions of the bacterial lawn around each disk. Measure and record the diameter and clarity of any cleared areas around the disks. Give quantitative measurements of your observations.
Filter Sterilization Procedure
- Attach pre-filter to syringe and “rinse” with water (H2O)
- Carefully open the sterilization filter while keeping the filter in its plastic covering
- Load approximately 1.5mL of water-based filtrate using a micropipet
- Depress the plunger, collecting the sterile-filtered filtrate into the microfuge tube
- Quickly close the microfuge cap until a “snap” sound is heard
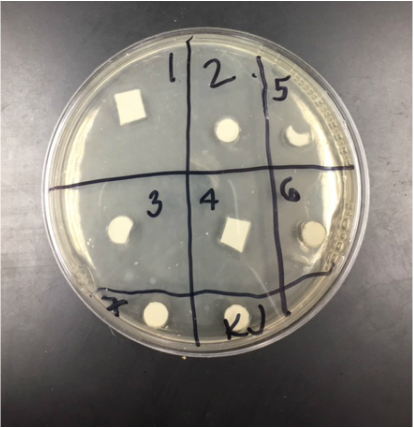
Results:
Our water saturated paper that was placed in the wrong area. There was no bacteria lawn and no bacterial prevention (the clearing that would have appeared around the paper shapes).
Our water saturated paper that was placed in the wrong area. There was no bacteria lawn and no bacterial prevention (the clearing that would have appeared around the paper shapes).
Analysis:
Our results were inconclusive, due to the misplacement of the water saturated paper square. If the paper had been in the proper area, more reasonable results would have been the outcome.
Our results were inconclusive, due to the misplacement of the water saturated paper square. If the paper had been in the proper area, more reasonable results would have been the outcome.